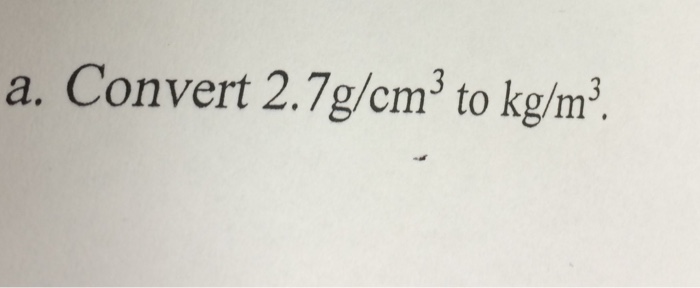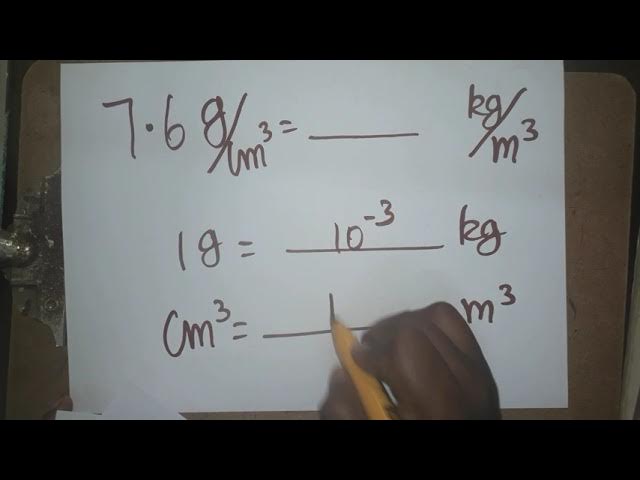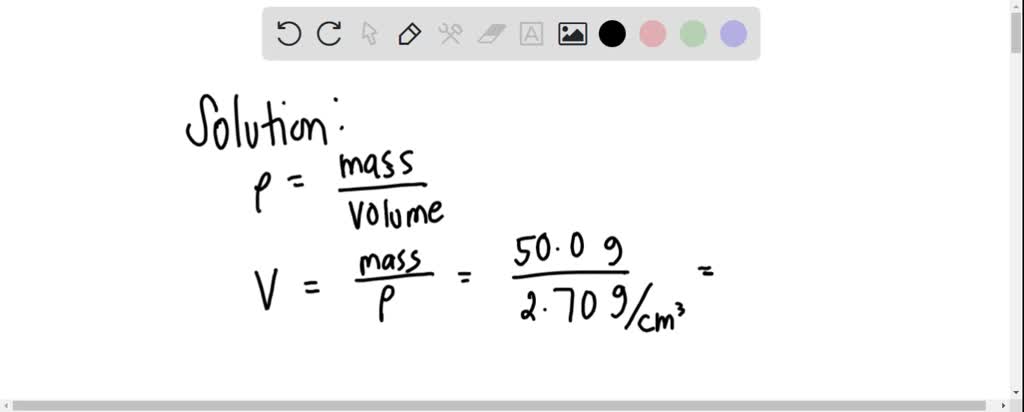
SOLVED: Density can be used as a conversion factor to convert between mass ( g) and volume (mL or cm3 ). Knowing that the density of aluminum is 2.70 g/ cm3 , calculate the

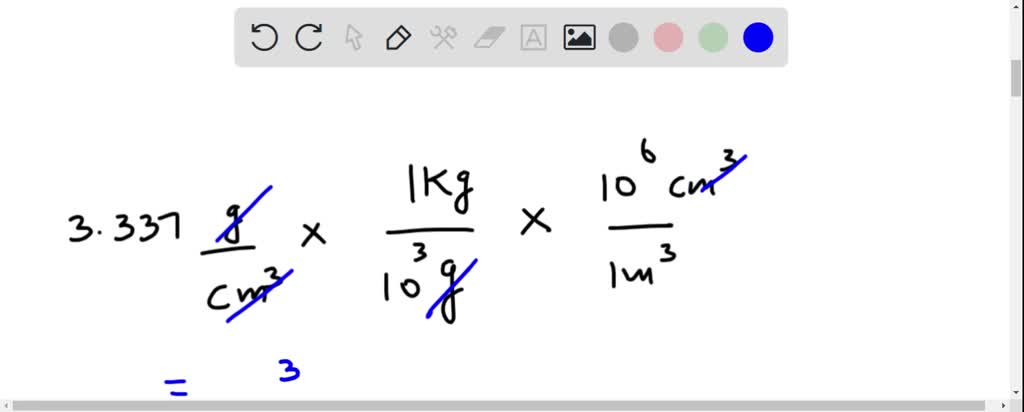
SOLVED: What is the unit conversion factor between density unit g/cm3 and kg/m3? Please show your work.