An Efficient Analytical Development Strategy for Rapid Development of Biopharmaceuticals | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Management of post-analytical processes in the clinical laboratory according to ISO 15189:2012. Considerations about the management of clinical samples, ensuring quality of post-analytical processes, and laboratory information management
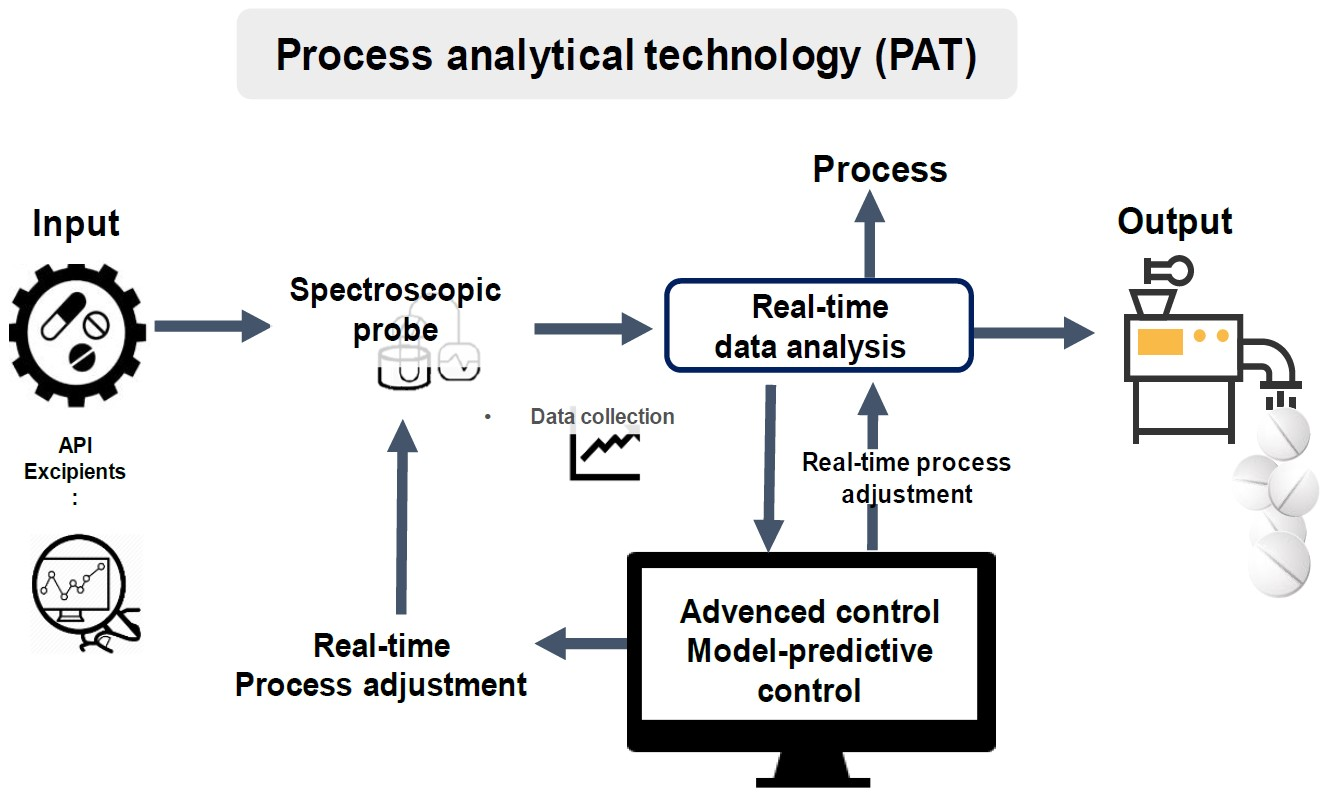
Pharmaceutics | Free Full-Text | Process Analytical Technology Tools for Monitoring Pharmaceutical Unit Operations: A Control Strategy for Continuous Process Verification

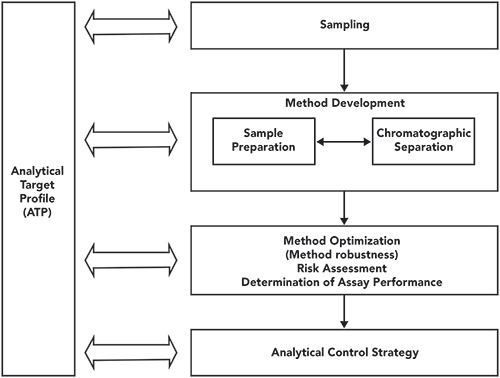
Unlocking the Power of Analytical Target Profile (ATP) and Analytical Control Strategy (ACS) - Keynotive
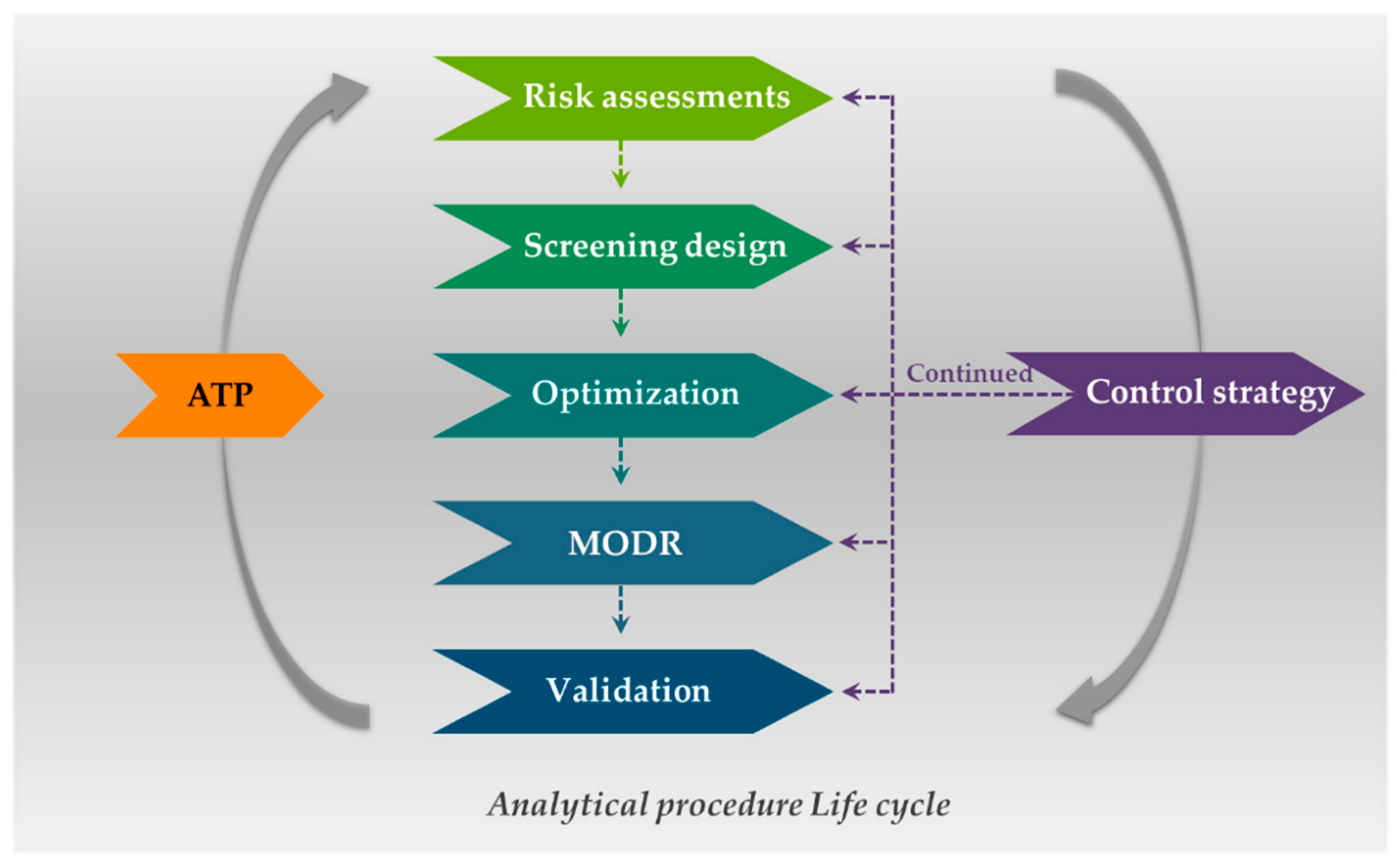
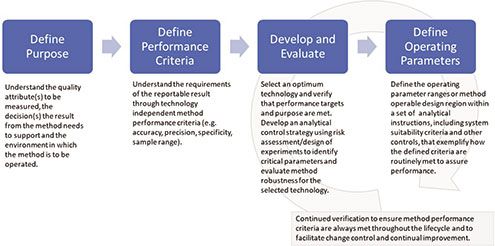
Plants | Free Full-Text | Analytical Quality by Design (AQbD) Approach to the Development of Analytical Procedures for Medicinal Plants

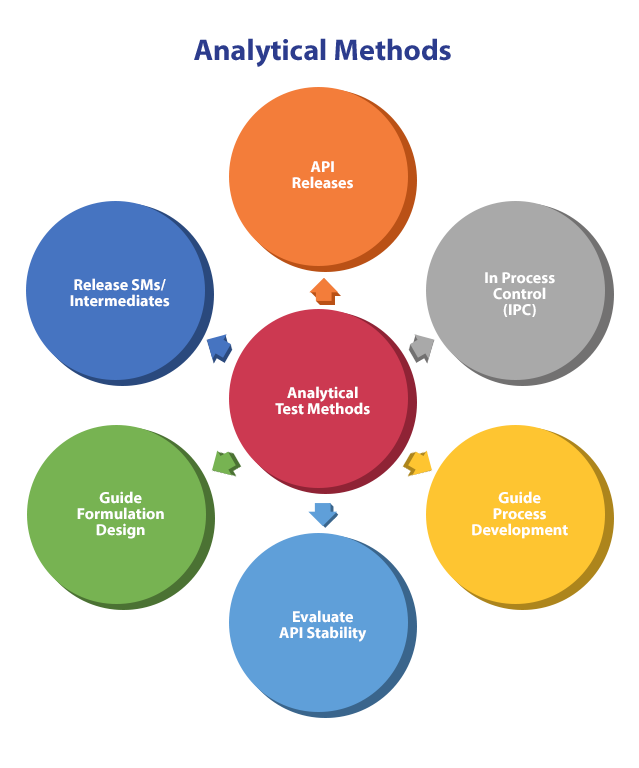
An Efficient Analytical Development Strategy for Rapid Development of Biopharmaceuticals | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology